Main content
Top content
Lab Techniques
Top content
1H and 13C NMR Spectroscopy
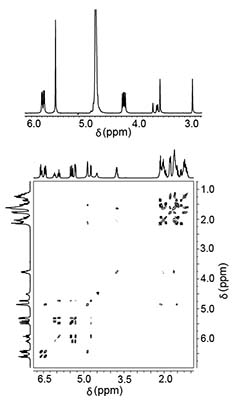
1H and 13C NMR spectra are essential for the structural determination of organic molecules and have to be measured for all newly synthesized compounds in our lab. Since the NMR chemical shift, δ, is also sensitive towards the environment surrounding the molecule, 1H NMR spectrosopy is also highly useful for the characterization of supramolecular host-guest complexes including the determination of their binding affinities.
More detailed information can be obtained using two-dimensional NMR spectroscopy. COSY, HSQC, and HMBC spectra are useful to elucidate the structure of more complex organic molecules. In supramolecular complexes, NOESY and ROESY experiments are used to confirm the spatial proximity of host and guest protons and EXSY and DOSY spectra give the exchange kinetics and diffusion coefficients of supramolecular structures.
Within our infrastructure, we have access to 500, 250, and 60 MHz NMR spectrometers including cryo probeheads, pulsed field gradient NMR (PFG-NMR), and magic angle spinning (MAS) accessory for solid-state NMR (ssNMR).
Optical Spectroscopy
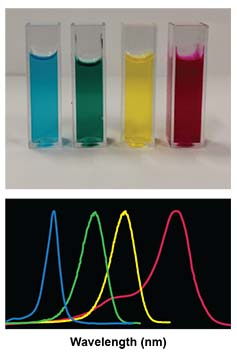
Absorption and fluorescence spectroscopy is used for the photophysical characterization of dye molecules, for example, for determining the absorption and emission maxima, the quantum yield, and the brightness of fluorescent dyes. Since the spectral properties of fluorescent dyes are influenced by molecular collisions and the micropolarity of the environment, interactions with biomolecules and supramolecules can be investigated in large detail using optical spectroscopy.
Various titration methods afford the number of binding sites, the affinity, and the stoichiometry of the binding interactions. Time-resolved measurements can be used to follow and determine the kinetics of exchange reactions and biochemical transformations.
Since fluorescence spectroscopic techniques are highly sensitive down to the single-molecule level, sophisticated fluorescence-based probes and sensors are highly demanded for fundamental and applied research as well as in automatized routine testing laboratories.
We have access to spectrometers for various optical spectroscopic characterization methods (absorption and fluorescence spectrometers with accessories für microplate measurements, polarization measurements, as well as stopped-flow accessory for fast kinetic measurements). Furthermore, we have access to the infrastructure of the Integrated Bioimaging Facility Osnabrück (iBiOS). Time-resolved fluorescence spectroscopic measurements with pico- and nanosecond time resolution are performed at external collaboration partners.
Nano- and Biomaterials

One of our goals is to create artificial, supramolecular system, which interact or become interfaced with bio- and nanomaterials. Consequently, we have experience with peptides, proteins, antibodies, enzymes, and DNA, as well as with gold and polymer particles. This includes bioconjugation methods, handling, quantification, and characterization of these materials, for which we have implemented various commercial and self-developed assay procedures.
For our membrane transport projects, we synthesize liposomes of various sizes from small to large and giant unilamellar vesicles (SUVs, LUVs, and GUVs), which can be prepared either with encapsulated molecules or empty, or as polarized vesicles with a membrane potential. Moreover, we have experience with various cell lines, for example, with mammalian chinese hamster ovary (CHO) cells and with various cancer cell lines (HeLa, HCT 116 und KTC-1). Within the CellNanOs, we have access to the respective cell culture labs.
Fitting Procedures, Simulations & Statistical Data Analysis
The functionality of our supramolecular systems is founded in interconnected binding and exchange equilibria. This ranges from simple host-guest binding, which can be solved analytically with high school mathematics, to more complex cases like competitive binding, allosteric control, and exchange mechanisms, which require numerical approximation methods. For a large variety of cases, we have developed in-house fitting procedures, which we further adapt to more complex problems.
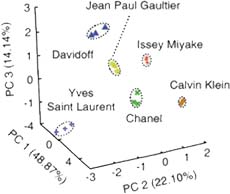
To improve the performance of our sensor systems, we also use various statistical multivariate data analysis methods, such as linear discriminant analysis (LDA), principal component analysis (PCA), and hierarchical cluster analysis (HCA).

