Hauptinhalt
Topinformationen
Macromolecular Chemistry
A series of functional macromolecules with different architectures has been prepared by means of free radical polymerization (FRP), Atom-Transfer Radical Polymerization (ATRP), and polymer analogous reactions. The materials have been investigated as surfactants, primer molecules, or for modification of surface polarity:
1. “Palm Tree” Polymers
“Palm Tree” Polymers are diblock copolymers consisting of a (long) block made from a low molecular weight monomer, and a second block consisting of a macromonomer or a copolymerizate of a macromonomer (see Scheme 1.1). Palm tree polymers can be prepared by sequential polymerization of the respective monomers. Amphiphatic Palm Tree polymers made from mutually immiscible blocks are interesting compounds because of their strong surface segregation tendencies. Due to their branched / multiarm nature these amphiphilic polymers strongly enrich at interfaces, and can hence be used as surfactants, micelle / vesicle generators or they can very effectively cover solid surfaces and help to modify surface properties.
![Scheme 1: Schematical depiction of a “Palm Tree” block copolymer P[A]-b-P[Macromonomer] Scheme 1: Schematical depiction of a “Palm Tree” block copolymer P[A]-b-P[Macromonomer]](/fileadmin/_migrated/pics/palmtreefigure001.jpg)
Schematical depiction of a “Palm Tree” block copolymer P[A]-b-P[Macromonomer] .
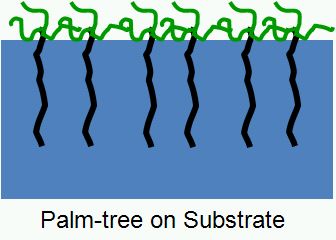
Scheme 1.1: Schematical arrangement of Palm Tree polymers at liquid interfaces.
2. “Double Comb” Polymers
“Double Comb” polymers are macromolecules with a linear backbone bearing two different kinds of polymeric side chains (see Scheme 2.1). Amphiphatic and amphiphilic double comb polymers can e.g. be made by copolymerization of macromonomers, by “grafting from”, as well as “grafting to” techniques. Similar to Palm Tree polymers these compounds tend to enrich at interfaces, and may be useful for surface modification purposes.
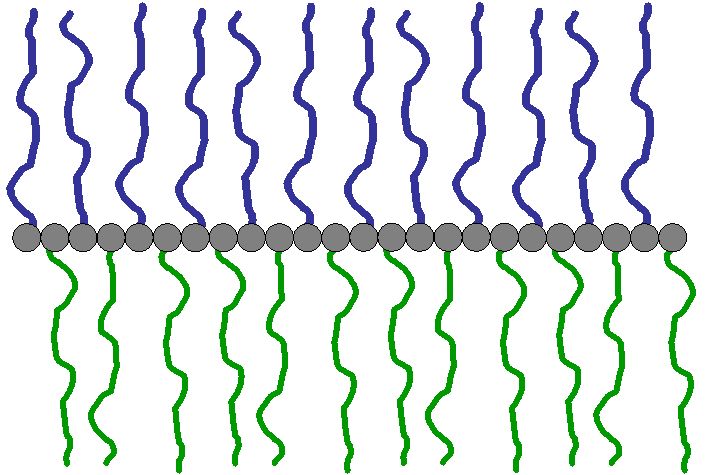
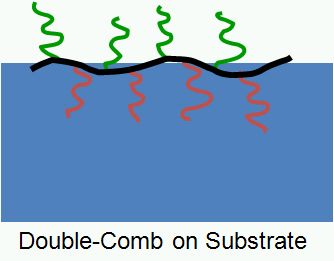
Scheme 2.1: Schematic depiction of a “Double Comb” polymer architecture and a possible arrangement of a Double Comb Polymer at a liquid interface
3. "Quat-Primer" Polymers
Present days surface treatments agents are “monodirectional” compounds that permanently adsorb on surfaces and specifically change the properties in the desired direction. For this reason the applied compounds must consist of moieties that bind to the surface and additional units that cause the property modifications. Because of this combination, for each surface and each application individual chemicals must be produced and kept in stock.
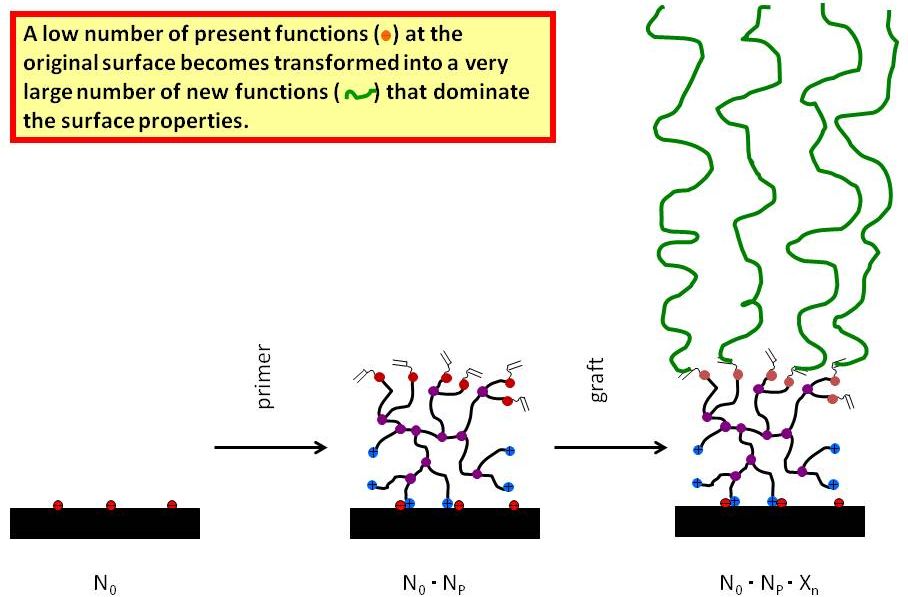
Figure 3.1: Functional group multiplication by Quat-Primer adsorption / polymerization
The aim of this project is to develop a technology that separates the elementary functions “linking” and “modification effect” and allows the generation of an “universal surface modification kit”. The surface treatment process is broken down in two independent steps: In the first step the substrate is furnished with a multi-reactive polymer that permanently binds to the substrate and forms a reactive platform that can be used to fix the desired modification agent in the subsequent step. Cationic compounds rapidly adhere to the majority of substrates and hence are frequently used as anchor groups to bind e.g. finishing agents on fabrics. Ultra thin layers of cationic polyelectrolytes were chosen as “connective platform” to bind finishing agents to textiles, carbon fibres or carbon nanoparticles. Such “polyquats” also contain reactive groups that do not react with the surface. On the polyquat - pre-treated substrates functional agents are bound in the second step that allow for the targeted control of surface properties (i.e. hydro- or oleophobicity, UV-protection, antimicrobial activity…). The technique offers a two step amplification of functional surface groups (cf. Figure 3.1).
Since the surface reactivity becomes unified by the polyquats, one and the same finishing agent can be applied to different materials. Furthermore the procedure offers the opportunity to divide a polyquat-treated material in different batches that can separately be finished with respect to different types of functionality [1].

Figure 3.2: Schematic depiction of polyethyleneimine based Amino-Quat-Primer Polymers at the interface between carbon fibers and epoxy resin matrix
Figure 3.2 schematically depicts the function of Quat-Primer Polymers to enhance the adhesion of epoxy resins on carbon fibres. Carbon fibres possess negatively charged surfaces allowing for very strong adhesion of quarternized macromolecules. The combination of positively charged- and epoxy reactive groups in one macromolecule offers a possibility to control the structure of the fibre / matrix interface and hence to improve the fibre / matrix shear strength [2].
Subsequent treatment of branched poly(ethylene imine) with N,N,N-trimethyl-3-({[(2-oxo-1,3-dioxolan- 4-yl)methoxy]carbonyl}amino)-propan-1-ammonium iodide and either (2-oxo-1,3-dioxolan-4-yl)methyl- (oxiran-2-ylmethyl)carbamate or 4-[(oxiran-2-ylmethoxy)methyl]-1,3-dioxolan-2-one yielded ‘quat-primer polymers’ bearing quarternary ammonium and cyclic carbonate groups covalently linked to the macromolecule. 1H and 13C NMR spectroscopy with model reaction mixtures and the macromolecular products revealed that the quaternary ammonium groups were selectively attached to the primary amino groups of the polymeric educt. The new functional macromolecules were characterized further by means of DSC and TGA. Upon spin coating the cyclic carbonate groups containing cationic polymers on oxygen/argon plasma-pretreated highly oriented pyrolytic graphite (HOPG), scanning force microscopy revealed the presence of a 3 nm thick film covering the substrate. This ultrathin coating was persistent against extraction with methanol, demonstrating the adherence of the polymer on the substrate. Applied on carbon fibers the polymer layer improved the interfacial shear strength between the carbon fiber and an epoxy resin matrix by 35% with respect to nontreated carbon fibers as demonstrated by single-fiber fragmentation tests [2].
Reference
- M. Moeller, U. Beginn, H. Keul, H. Thomas, “Method for producing adherent coatings to solid surfaces.”, DWI an der RWTH Aachen e.V., Germany, Eur. Pat. Appl. (2006), 20pp., EP 1710282 A1 20061011 Application: EP 2005-7306 20050404.
- V. Goel, U. Beginn, A. Mourran, M. Möller, “‘Quat-Primer’ Polymers bearing cationic and reactive groups: synthesis, characterization and application”, Macromolecules 41, 8187-8197 (2008)
4. Gradient Copolymers
Gradient copolymers represent a relative new class of copolymers containing at least one section of the macromolecule where the chemical composition continuously changes along the polymer chain (cf. Figure 4.1). Due to their structural relation to block copolymers, microphase-segregated structures can be formed in bulk, in solution, at surfaces, and interfaces exhibiting a blurred, less-defined border region between phases [1]. Actual research is directed towards (i) the development of effective preparation techniques and (ii) the evaluation of possible applications for these new class of polymers.
![Scheme 4.1: Schematic depiction of the monomer sequences in a diblock copolymer P[A]-b-P[B] (a), a gradient copolymer P[A-grad-B] (b), and a random copolymer P[A-co-B] (c) of equal composition (50 mol% A) Scheme 4.1: Schematic depiction of the monomer sequences in a diblock copolymer P[A]-b-P[B] (a), a gradient copolymer P[A-grad-B] (b), and a random copolymer P[A-co-B] (c) of equal composition (50 mol% A)](/fileadmin/_migrated/pics/gradientpolymers001.jpg)
Scheme 4.1: Schematic depiction of the monomer sequences in a diblock copolymer P[A]-b-P[B] (a), a gradient copolymer P[A-grad-B] (b), and a random copolymer P[A-co-B] (c) of equal composition (50 mol% A)
Two experimental approaches have been distinguished to control the compositional gradient, namely the ‘‘spontaneous’’-and the ‘‘forced’’-gradient methods. Spontaneous gradient polymerizations are batch-copolymerization reactions where a monomer mixture is polymerized to high conversions. In the absence ofeither azeotropic points or ideal random copolymerizations the monomer composition will change gradually with the monomer conversion because of the different reactivities of the monomers. As determined by Skeist’s equation a compositional gradient evolves along the polymer chain. This technique is quite simple, but suffers from three drawbacks: it is only applicable to strictly non-random copolymerization systems and will fail in close vicinity to azeotropic points; it cannot be used to cross the full compositional space from F1 = 0 to F1 = 1 and it is virtually impossible to generate constant compositional gradients over the full range of monomer composition.
![Scheme 4.2: Schematic depiction of the composition of a P[M1]n-b-P[M1-grad-M2]Xe-b-P[M2]m gradient triblock copolymer. Scheme 4.2: Schematic depiction of the composition of a P[M1]n-b-P[M1-grad-M2]Xe-b-P[M2]m gradient triblock copolymer.](/fileadmin/_migrated/pics/gradientpolymers002.jpg)
Scheme 4.2: Schematic depiction of the composition of a P[M1]n-b-P[M1-grad-M2]Xe-b-P[M2]m gradient triblock copolymer.
‘‘Forced’’-gradient copolymerizations are semi-batch reactions involving the initiation of a polymerization in a pure monomer or a monomer mixture and the subsequent continuous addition of a second monomer or a monomer mixture during the reaction time. The addition rate as well as the composition of the added mixture may be varied to allow for full control on the composition of the generated copolymer chain. Block copolymers of the structure P[M1]n-b-P[M1-grad-M2]Xe-b-P[M2]m containing constant compositional
gradients with composition ranges within the gradient block from F1 = 1 to F1 = 0 (see Scheme 4.2) can only be prepared by means of forced gradient techniques [1,2].
The ongoing project is directed towards the rational synthesis of constant gradient (block)copolymers by means of continuous addition [3] controlled radical polymerization. In particular a simplified scheme was developed that allows to prepare well defined gradient copolymers with a minimum of experimental efforts and low equipment requirements [2]. In case that an arbitrary gradient function g(X) shall be realized, one has to solve the differential equation system (DES) (Eqs. (1) and (2)) to calculate the amount of monomer to be added at a certain monomer conversion p.
Eq. 1)
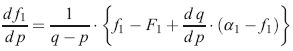
Eq. 2)
(g(X)=targeted copolymer compositional gradient, Xe=targeted length of the gradient block, F1‘=dF1/df1, q=total monomer addition function, a1=molar fraction of monomer 1 in added monomer mixture, p=monomer conversion, F1= instantaneous molar fraction of monomer 1 in the copolymer, f1= instantaneous molar fraction of monomer 1 in the monomer mixture).
To make the scheme work, the time dependency of the monomer conversion, as well as the required monomer addition is needed. The complete description of polymerization mechanisms requires a large number of kinetic parameters that may sometimes be cumbersome to retrieve. For this reason, a simplified ‘phenomenological’ approach has been proposed [2] that replaces the details by an empiric ‘gross’ kinetic function (cf. Eq. (3)), and extends the DES (Eqs. (1) and (2)) to predict the conversion/time relation. Note that Eq. (3) is an approximation that does not take into account the volume contraction of the solution due to polymerization.
Eq. 3)
(p=monomer conversion, t=reaction time, k(f1)·[I]0=effective kinetic rate constant, f1=molar fraction of monomer 1 in the solution, q=monomer addition function, y=dilution factor due to monomer addition).
The required parameter function to control a specific binary copolymer system, k(f1), can be collected by means of a low number of pilot polymerizations. The scheme makes a compromise between the full theory or full equipment-based ‘perfect control procedures’ on one hand and the ‘try and error’ constant rate addition programs on the other hand. The procedure was tested with the ATRP copolymerization of the almost ideal randomly copolymerizing pair n-butyl methacrylate/t-butyl methacrylate to yield satisfying control up to monomer conversions of 74–87%, although the dilution effect was not taken into account (Eq. (3); y=0).
References
- U. Beginn, “Gradient Copolymers”, Coll. Polym. Sci. 286, 1465-1474 (2008).
- U. Beginn, “Monomer addition programs to generate constant gradient block copolymers”, Polymer 47(19), 6880 – 6894 (2006).
- U. Beginn, “Continuous Addition Polymerization”, e-Polymers, E_003, 1-109 (2005).
5. Anhydride reactive Copolymers
This subject is presently under construction.
6. Reactive Graft Copolymers by Copolymerization of Macromonomers
Graft- or Comb copolymers are macromolecules bearing macromolecular side chains covalently attached to their polymeric backbone. Graft copolymers can be made by “grafting-from” (i.e. polymerization of the side chain starting from an initiator moiety fxed to the backbone), “grafting-to” (attachment of ready-made polymer chains to a backbone) or by homo- or copolymerization of macromonomers. The research in this project group of the department is directed towards the preparation of polymers with poly(dimethylsiloxane) (=PDMS) side chains by copolymerization of PDMS-mono methacrylate (PDMS-MA). As depicted in Scheme 6.1 the use of free radical polymerization will produce graft polymers with random distributions of graft chains along the backbone, while controlled polymerization techniques will yield “Palm tree” block copolymers.
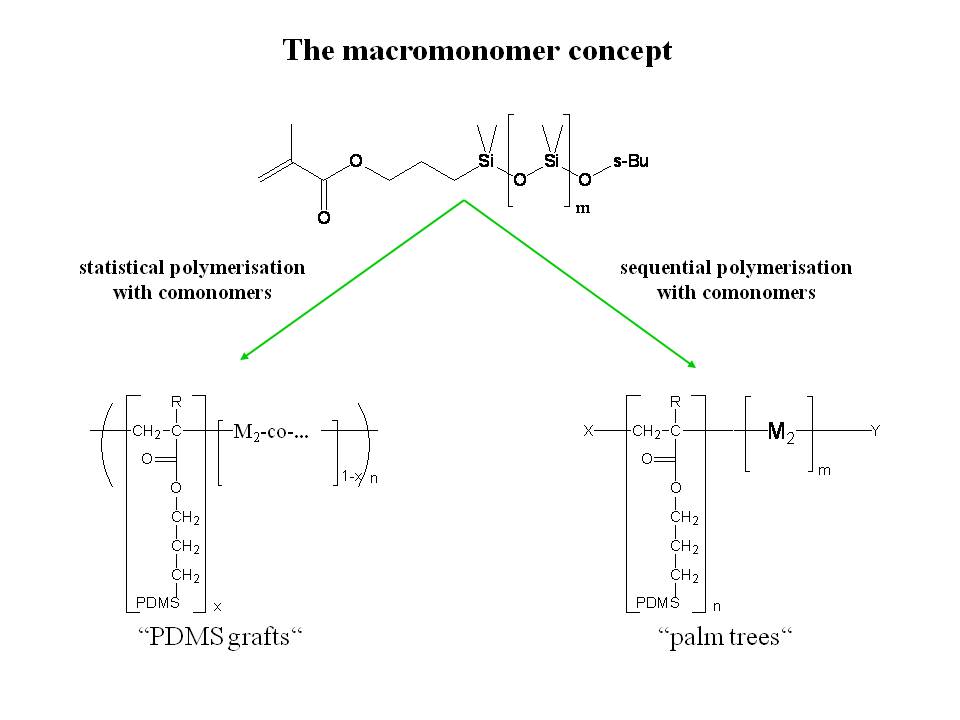
Scheme 6.1: Preparation of Graft-Copolymers by copolymerization of macromonomers
Copolymerization of PDMS macromonomers with suitable - preferrably reactive - monomers, such as anhydride monomers, alkoxysiloxane-monomers or functional (meth)acrylates results in functional / reactive PDMS graft copolymers (see Scheme 6.2) gives facile access to functional PDMS graft copolymers.
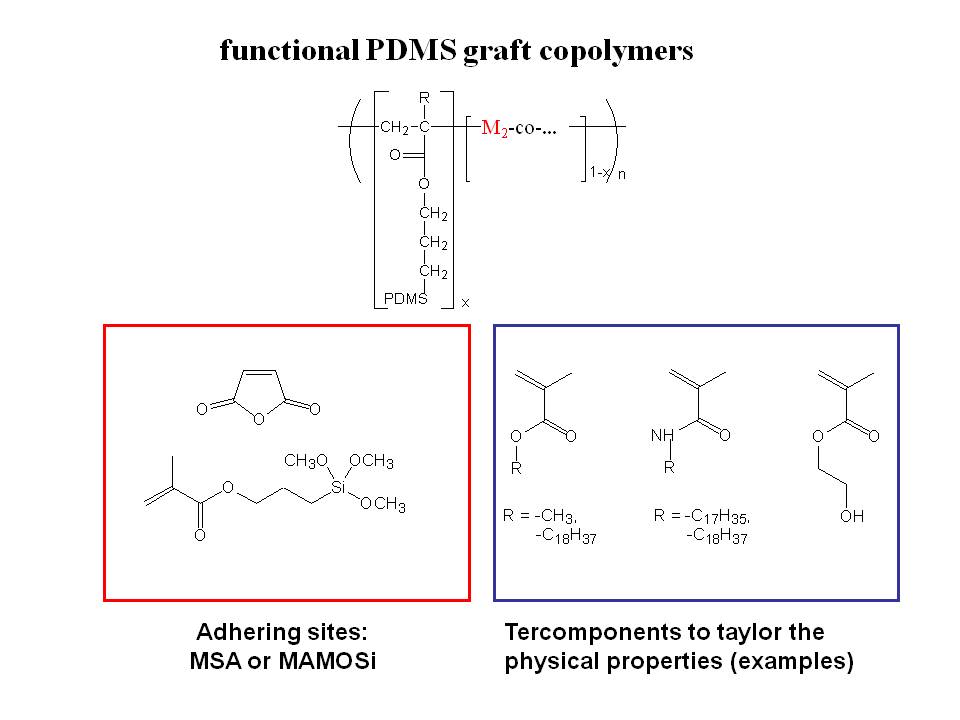
Scheme 6.2: Prepared PDMS-Graft-Copolymers by copolymerization of PDMS-MA macromonomers
The use of anhydride monomer units in the backbone allows for polymer analogous reactions under imide-ring formation, opening the pathway towards e.g. "Double Comb" copolymers (Scheme 6.3).
![Scheme 6.3: Polymer analogous modification of P[PDMS-MA-co-MSA] copolymers Scheme 6.3: Polymer analogous modification of P[PDMS-MA-co-MSA] copolymers](/fileadmin/_migrated/pics/macromonomercopo003.jpg)
Scheme 6.3: Polymer analogous modification of P[PDMS-MA-co-MSA] copolymers

